Mayo Clinic is taking epilepsy research in a bold new direction, exploring treatment approaches to help patients living with the most severe and difficult-to-treat forms of epilepsy. About 50 million people worldwide are impacted by epilepsy. Approximately 30% of patients, or about 15 million people, suffer with drug-resistant epilepsy (DRE). While some patients experience only a few seizures per month, others may endure hundreds each day - ranging from episodes that are mild to life-threatening.
Current treatment options for patients with DRE include surgical procedures such as brain resection to remove a portion of the brain tissue responsible for generating seizures. A less invasive procedure involves laser ablation therapy that pinpoints and destroys abnormal brain tissue. While often effective, these surgical approaches carry the risk of possible side effects, such as memory impairment, motor deficits and speech difficulties. Neuromodulation is another surgical approach that uses electrical or magnetic stimulation to interrupt abnormal neural activity without removing brain tissue.
Now, a growing number of scientists across the globe are part of an innovative trend in research investigating novel ways to DRE. It involves the use of regenerative medicine as a "reparative" approach to help the brain heal.
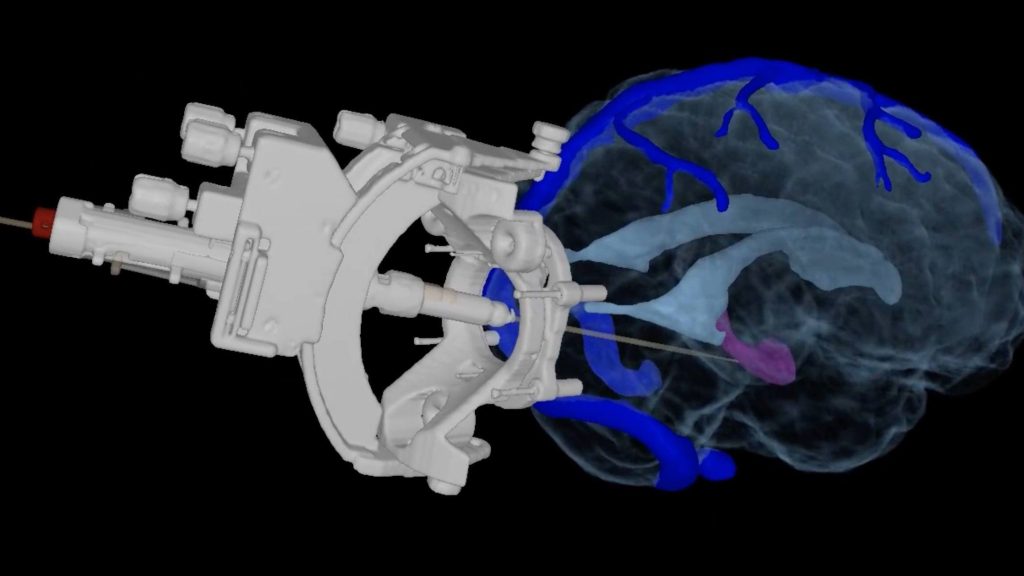
Neurosurgeon Jonathon J. Parker, M.D., Ph.D., is the lead investigator of the first-in-human clinical trial at Mayo Clinic studying the use of implanted specialized inhibitory brain cells as a potential reparative treatment for DRE. The clinical trial is underway at Mayo Clinic in Arizona.
"This is an exciting time for regenerative medicine and the potential it may have for millions of people who suffer from the debilitating side effects of DRE," says Dr. Parker. "We use a very minimally invasive technique where we inject the inhibitory cells through a pencil eraser-sized incision in the back of the head. Our hope is that, over time, these cells become part of the brain and help repair the neural circuitry, and reduce or prevent seizures without the side effects," says Dr. Parker.
Mayo Clinic in Arizona is one of 29 sites nationwide participating in the inhibitory brain cell implant clinical trial for patients with focal epilepsy, where seizures originate in a specific region of the brain.
Arizona resident Anthony Maita was the first person at Mayo Clinic to participate in the clinical trial. He underwent the one-time, single-dose procedure and was discharged from the hospital the next day. "I had no trouble with it," says Anthony. "My biggest hope is that, one day, I don't have to deal with this. My other biggest hope is that other people won't have to either."
It is still too early to determine whether the brain cell implant was effective for Anthony. Doctors are monitoring his progress closely. "Anthony has been doing great since the procedure," says Amy Crepeau, M.D., neurologist at Mayo Clinic. "We have a great deal of optimism in regard to the potential of this brain cell therapy. Developing a safe and effective, minimally invasive treatment that does not carry the possible negative side effects could be a game changer in treating patients with DRE and improving their quality of life."
Another clinical trial is underway at Mayo Clinic in Florida investigating the potential of regenerative medicine as a reparative treatment for DRE. Researchers are exploring the use of implanted stem cells in conjunction with neuromodulation.
One of the most recent FDA-approved methods of neuromodulation therapy for epilepsy is deep brain stimulation. While patients who undergo deep brain stimulation experience median seizure reduction up to 70% after five years, it is uncommon for patients to become seizure-free. Sanjeet Grewal, M.D., director of stereotactic and functional neurosurgery at Mayo Clinic, is hoping to change that. "Unfortunately, neuromodulation doesn't give us the seizure freedom we want, and that's why we are trying to combine deep brain stimulation with stem cell therapy to see if we can increase the efficacy of neuromodulation," says Dr. Grewal.
Dr. Grewal is the lead investigator of the clinical trial which involves the use of implanted adipose-derived mesenchymal stem cells (MSCs) as an adjunct to deep brain stimulation for DRE patients. MSCs are a special type of adult stem cell with anti-inflammatory properties that may also have potential for healing.
Many, like Dr. Grewal, hope MSCs will serve a pivotal role in the future of regenerative medicine to treat conditions like epilepsy. "There are some patients whose seizures are just much harder to treat with the technology we have today. Our hope is that by adding stem cells and their regenerative potential, we can increase treatment success," says Dr. Grewal.
The clinical trial is using MSCs derived from fat tissue and produced at the Human Cell Therapy Laboratory at Mayo Clinic in Florida under the leadership of Abba Zubair, M.D., Ph.D. His research teams have developed a cost-effective method of producing MSCs for use in potential treatments for conditions such as stroke and osteoporosis. "My mission is to discover ways to address problems that patients have been struggling with and find a solution for them. I want to give them hope," says Dr. Zubair. "I truly believe the future is bright."
"MSCs are what we call multipotent, meaning they can differentiate into different cell types based on where they're placed. If they are placed near blood vessels, they can become blood vessel types. If they're placed by heart cells, they can become heart cell types," explains Dr. Grewal. It is hoped the MSCs in Dr. Grewal's clinical trial will become neural or brain cell types and interact in the part of the brain where seizures occur. "It's called paracrine signaling, where they're releasing signals to the brain tissue around them and interacting in a way to try to repair that tissue."
Tabitha Wilson began having seizures at the age of 2. The Florida resident says her seizures were well controlled until her mid-20s when her medication stopped working. Tabitha tried numerous other medications and underwent three brain surgeries, none of which provided the relief she needed.
"There are days when I'll have two, three or four seizures, back-to-back," says Tabitha. "I fell down a flight of stairs. I've burned myself while cooking. I've completely blacked out and don't know where I am." Like many people who have epilepsy, Tabitha says uncontrolled seizures have robbed her ability to live independently. "I can't drive, can't cook or swim alone. I can't take a bath, only a shower and someone has to be in the house," says Tabitha.
Tabitha became the first person to participate in the Florida clinical trial. Dr. Grewal says she is also the first person in the world to undergo surgery for deep brain stimulation and receive stem cell therapy in the thalamus in her brain as a potential treatment for DRE. "I'm willing to try everything and anything to get some sort of control over these seizures because I've been living with this for so long," says Tabitha. "I hope to be a mother someday."
Since the surgery, Dr. Grewal says there has been an improvement in Tabitha's seizure management. However, he says it is too early to know whether this is due to the deep brain stimulation, stem cells or both.
Drs. Grewal and Parker say there is a long road ahead to determine whether these cell therapies are proven safe and effective for patients with DRE. But they agree each day brings them one step closer to a potential treatment or even a cure for patients like Tabitha and Anthony.
"We've thought about this for generations, we just didn't have these technologies to enable it. Now we do," says Dr. Grewal. "So, whether it's wound healing, neurodegeneration, epilepsy or stroke, there are so many different studies going on investigating the potential of regenerative or reparative therapies."






