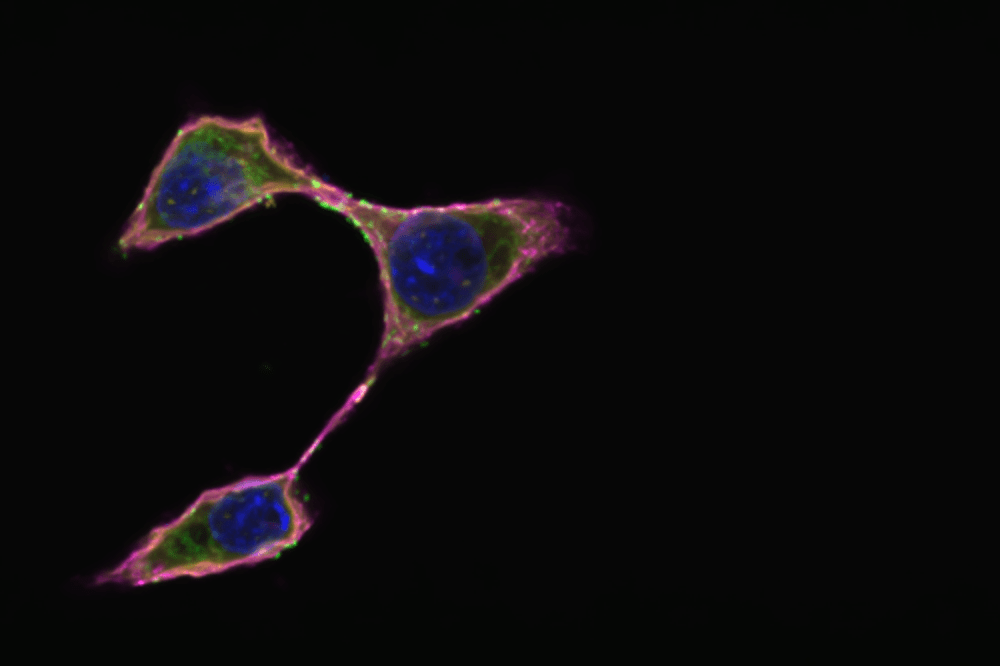
Glycosphingolipids (in pink, via Cholera Toxin B) are so critical for cancer immune evasion that certain cancer cells cannot proliferate without it.
Cancer cells seldom start off stealthy. Quite to the contrary, they announce their presence to the immune system by planting chemical red flags right on their membranes. Once alerted, the body's defenses can swoop in, destroying rogue cells before they can do much damage. Lying at the heart of this early warning system are lipids, fatty compounds previously seen by cancer biologists primarily as a fuel source for burgeoning tumors.
But now, a new study in Nature demonstrates that one particular lipid type is actually critical for cancer immune evasion-so much so that certain cancer cells cannot proliferate without it. The findings confirm longstanding suspicions that not only is this lipid a key player in cancer biology (and therefore a key drug target), but also demonstrate that existing FDA-approved medications designed to stunt lipid production can galvanize the immune system against cancer.
"Cancer cells are altering how this lipid is metabolized, which in turn distorts the 'eat me' signals that malignant cells usually produce," says first author Mariluz Soula, a former graduate student in the laboratory of Kivanç Birsoy, and now a scientist Lime Therapeutics. "This paints a very different picture of the role lipids play in cancer growth."
A mysterious connection between lipids and cancer
Scientists have long known that cancer cells alter lipid metabolism, but it was generally assumed that cancer cells were gobbling up these lipids for energy-consuming the fatty molecules to help the tumor grow and spread far beyond that of healthy cells.
"We knew from the literature that elevated lipid levels correlate with severity of cancer growth and metastasis, but it was unclear how," Soula says. The Birsoy lab, in conjunction with the laboratory of Gabriel D. Victoria, set out to answer this question by screening the genes involved in this process. They then implanted a series of cancer cells, each missing a different such gene, into mice with and without immune systems-thereby revealing which lipids a cancer cannot live without.
The result: so-called "sphingolipids." Discovered in the late 1800s by German chemist Johann Ludwig Wilhelm Thudichum, sphingolipids were named after the enigmatic Sphinx of Greek lore because of their puzzling structure and function. Two centuries later, sphingolipids are less of a mystery. "We know that sphingolipids aren't really used for energy," Soula says. "They're mainly in the cell membrane to create scaffolding for signaling proteins."
This finding raised an intriguing possibility. Was lipid metabolism in cancer cells really just the story of hungry cells trying to consume more energy? Or was it a key part of the cancer cell's efforts to subtly manipulate cell signaling and dodge the immune system?
Toward a new therapeutic strategy
To test how sphingolipids were driving cancer growth, the team turned to an FDA-approved drug used to treat Gaucher disease-a genetic disorder characterized by an impaired ability to break down lipids. The drug essentially blocks glycosphingolipid synthesis, and the team found that this impaired tumor growth in pancreatic, lung, and colorectal cancer models.
They also found that depleting glycosphingolipids prevented the formation of the "lipid nanodomains" that bunch signalizing molecules together on the membrane, impacting the cell's surface receptors on the cell surface in a way that made them more sensitive to an immune response. These findings suggest that cancer cells hoard glycosphingolipids in order to obscure inflammatory signals, and that disrupting glycosphingolipid production can leave cancer cells vulnerable to the immune system.
"Everyone thought of elevated lipid levels as an energy source for cancer cells to consume," Soula says. "We discovered that it's far more nuanced. Lipids are not just fuel, but a protection mechanism for cancer cells that modulates how they communicate with the immune system."
Future work will determine whether this holds true for multiple cancers. The team tested a variety of types, but found this mechanism at work in KRAS-dependent cancers (so named for the mutated oncogene that drives them). Still, the initial results could have significant clinical impact, given how aggressive many KRAS-dependent cancers, such as pancreatic cancer, tend to be. Based on their findings, the team suggests that drug and dietary interventions that stunt sphingolipid production may help increase the efficacy of existing immunotherapies.
"Diets may impact many aspects of cancer biology," Birsoy says. "We believe modulating dietary lipids may be an interesting avenue to target cancer cells' ability to evade immune cells."






