Organogold(III) complex accumulates in mitochondria of lung cancer cells
Precious metals are not merely ornaments; they are also important components of pharmaceuticals, like the antitumor drug cisplatin. Recently, the search for alternatives with improved activity has begun to focus on gold. In the journal Angewandte Chemie, a French research team has now published the first study about the speciation and distribution of an organogold(III) complex in cancer cells and reveals how specially designed "organogold" complexes might open exciting avenues for fighting cancer.
© Wiley-VCH, re-use with credit to 'Angewandte Chemie' and a link to the original article.
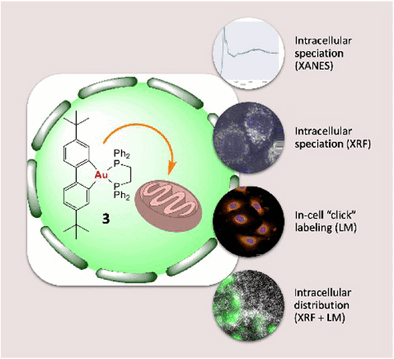
Gold has a unique electronic structure giving it exceptional chemical traits that translate into subtle interactions with biological molecules. Yet, to date, we have little information about how gold(III) complexes with antitumor activity behave in a biological environment. Do they change? Are they reduced to gold (I) or metallic gold? Where in the cell do they attack? Researchers led by Benoît Bertrand, Michèle Salmain, Sylvain Bohic, and Jean-Louis Hazemann at Sorbonne Université, the Université Grenoble Alpes, CNRS, INSERM, and the European Synchrotron Research Facility, have now carried out a comprehensive study on the chemical reactivity and antitumor activity of various gold(III) complexes. They used a combination of different methods based on synchrotron X-ray radiation-very intensive, bundled flashes of light produced in particle accelerators.
Common to all the complexes they examined (cationic biphenyl gold(III) complexes with aryl, alkyl, and diphosphine helper ligands, known as [(C^C)Au(P^P)]+ cations) is a gold atom bonded to two carbon atoms of the first ligand and two phosphorus atoms of the second, clasping like two sets of tongs. The analyses demonstrate that all the complexes examined were stable in both cell-free environments and inside lung cancer cells. They were not reduced and did not release their ligands to form new bonds.
The complexes were demonstrated to be toxic against tumor cells. A "dppe complex" (biphenyl gold(III) complex with 1,2-diphenylphosphinoethane (dppe) ligand) was the most active. The team used a special setup of synchrotron cryo-X-ray nanoanalysis to "map" elements including gold in frozen-hydrated lung cancer cells with nanometer-scale resolution and locate the dppe complex. It was found to accumulate selectively in the mitochondria, the "powerhouses" of the cells. The advantage of this method is that no labeling, which could distort the result, is needed. This gives scientists a unique clarity when examining cells in their near-native state at the nanoscale.
By using X-ray absorption spectroscopic methods, the team obtained important information about the valency, geometry, and oxidation state of the gold atom in the complex. These indicate that the antitumor activity of the gold complexes primarily stems from the native cationic species (the [(C^C)Au(P^P)]+ cations). It probably results from interactions between the whole complex and specific biological molecules, whose function is disrupted. This differentiates these drug candidates from other, differently structured gold complexes, which generally trigger cell death through direct coordination of the gold center with biomolecules. These results establish a relationship between the chemical structure and reactivity of a gold complex, its speciation in the cell, and its cytotoxicity.
(3419 characters)
About the Author
Dr. Benoît Bertrand is a CNRS junior researcher at the Institut Parisien de Chimie Moléculaire at Sorbonne Université. His research interest is the development of new organometallic gold complexes and the study of their anticancer properties with a particular focus on the intracellular reactivity of the complexes.
Copy free of charge-we would appreciate a transcript/link of your article. The original articles that our press releases are based on can be found in our online pressroom.






