As the saying goes, two wrongs don't make a right. But can two rights cancel out a wrong?
It can when the right things are hydrogen and carbon-two resources packed with energy-and the wrong thing is carbon dioxide (CO2), a notorious greenhouse gas.
Researchers from Pacific Northwest National Laboratory (PNNL) and West Virginia University (WVU), in collaboration with industry partners Southern California Gas Company (SoCalGas) and C4-MCP, have developed a process that converts methane-the primary component of natural gas-into hydrogen while emitting zero CO2. The process also creates carbon solids for manufacturing applications.
The hydrogen can be used in fuel cells for transportation including trucks, and large-scale energy storage, while the high-quality carbon products are suitable for a wide range of manufacturing applications, such as electronics, medical devices, aerospace composite materials, and building systems. Commercial sale of the carbon products offsets the cost of hydrogen production-a critical factor for industry.
"Hydrogen fuel will be a necessary component in achieving carbon neutrality, so we have to figure out ways to produce it cleanly and economically," said Ron Kent, technology development manager for low carbon resources at SoCalGas. The company co-sponsored the research along with C4-MCP, LLC, and the U.S. Department of Energy's (DOE) Hydrogen and Fuel Cell Technologies Office within the Office of Energy Efficiency and Renewable Energy. "Although this new method is still in the early stages, the results of the lab-scale tests look super promising," Kent said.
The novel "blue hydrogen" approach-clean hydrogen from natural gas versus renewable resources-could help California achieve its goal of cutting greenhouse gas emissions by 40 percent from 1990 levels. As part of that goal, the state aims to replace five million standard gas-powered vehicles with low- or zero-emission vehicles by 2030. Those targets will also help the state meet health-based air quality requirements established in the federal Clean Air Act.

Growing carbon from nickel
For decades, PNNL scientists and engineers have led research using catalysts to reduce CO2 emissions from industrial processes and transportation. Former PNNL research engineer, John Hu, now an endowed chair professor of the Statler College of Engineering and Mineral Resources at WVU, was one of those engineers.
Hu experimented with catalysts and processes that could cleanly convert methane-the primary component of natural gas-into both hydrogen and carbon during a process known as pyrolysis. During pyrolysis, a gas or liquid is cycled over a solid catalyst material under high pressure and temperature inside a closed vessel. The ensuing chemical reaction transforms the material's basic properties into other valuable fuels and products.
Continuing his research at WVU, Hu discovered a nickel-based catalyst formulation that stayed anchored to its support structure while growing carbon nanocrystals. This anchoring could enable the recovery of pure carbon nanotubes (CNT) and nanofibers, as well as catalyst regeneration.
"The purity and crystallinity of carbon products made from natural gas is important," said Hu. "The carbon products are not necessarily sold just in existing markets. They can be further upgraded to penetrate a number of markets where carbon products are currently made from petroleum."
Hu's results, published in 2017 in Catalysis Science and Technology, caught the attention of PNNL and industry, and formed the basis for the national laboratory-university-industry project through funding from the DOE Office of Energy Efficiency and Renewable Energy's H2@Scale initiative for clean hydrogen use across the economy.
Putting the catalyst to the test
The team's first step was to perform advanced catalyst tests and thoroughly characterize the resulting carbon products. Rob Dagle, a chemical engineer who had worked with Hu on past research projects, led the testing and analysis at PNNL, home of the Institute of Integrated Catalysis. The team also took advantage of scientific expertise and capabilities at the Environmental Molecular Sciences Laboratory (EMSL), a DOE scientific user facility located on the PNNL campus.
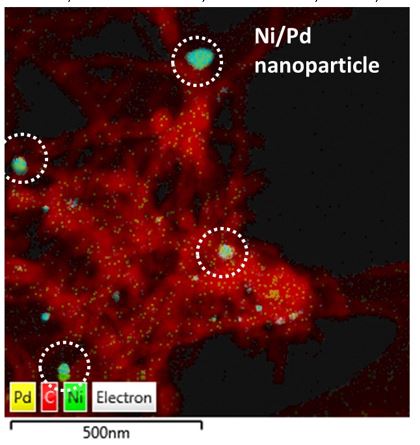
The researchers found that controlling the growth mechanism of CNTs was very difficult. To achieve the desired growth type, catalyst improvements were needed. Facing the challenge head on, the team didn't give up, but doubled down.
Through systematic testing of different-sized nickel particles paired with other metals, a clear picture emerged. The addition of a second metal altered the mechanism of carbon growth and further improved catalyst stability.
"The key was the size and structure of the nickel particle," said Dagle. "We quantified that catalyst stability increases with particle size. Also, if the nickel is too small, other types of carbon form and completely cover the catalyst, blocking the activity instead of growing into these nice long crystalline nanotubes."
Images and analysis from EMSL's scanning electron microscope confirmed the particle structure and metal dispersion on the catalyst surface. Another EMSL instrument, a transmission electron microscope, verified the internal structure and texture of the fresh catalysts, spent catalysts, and the carbon products.
The researchers then developed and demonstrated a thermocatalytic decomposition (TCD) method to separate and resynthesize the catalyst for a closed loop process. The team's research, "Structure sensitivity and its effect on methane turnover and carbon co-product selectivity in thermocatalytic decomposition of methane over supported Ni catalysts," appeared in Applied Catalysis A in December 2020.
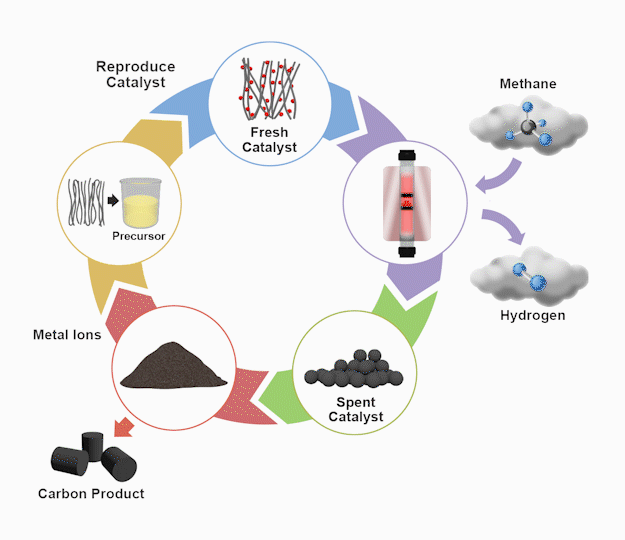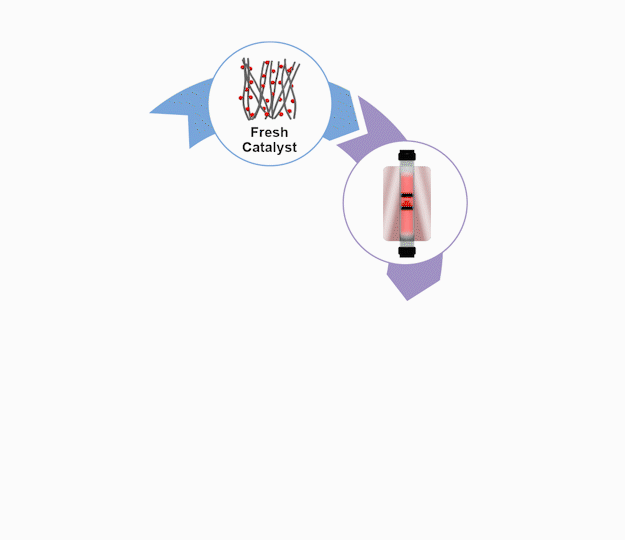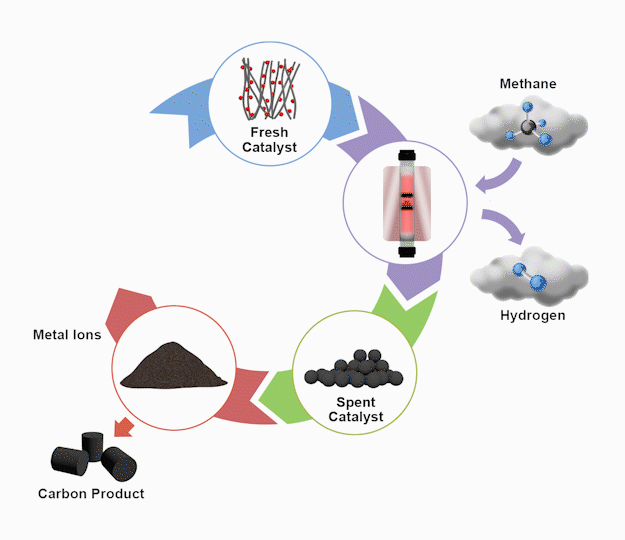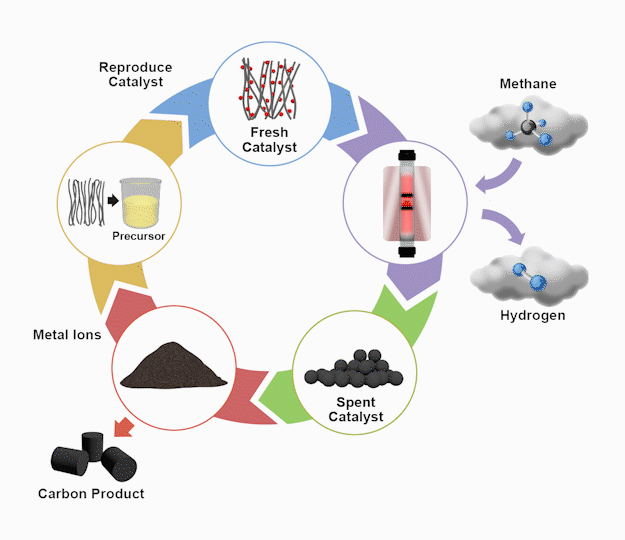
By the numbers
The TCD process starts by flowing methane gas through a patent-pending bimetallic catalyst inside a reactor vessel operating at approximately 600oC. The chemical reaction produces hydrogen as solid carbon accumulates on the catalyst. After this, an acid wash separates the carbon products from the metallic catalyst precursors. Next, the precursors are re-synthesized using some of the carbon product as the catalyst support. As the final step, the recycled catalyst re-enters the reactor, completing the loop for continuous catalyst replacement. The high-value carbon products gleaned from the wash process remain separate for industrial use.
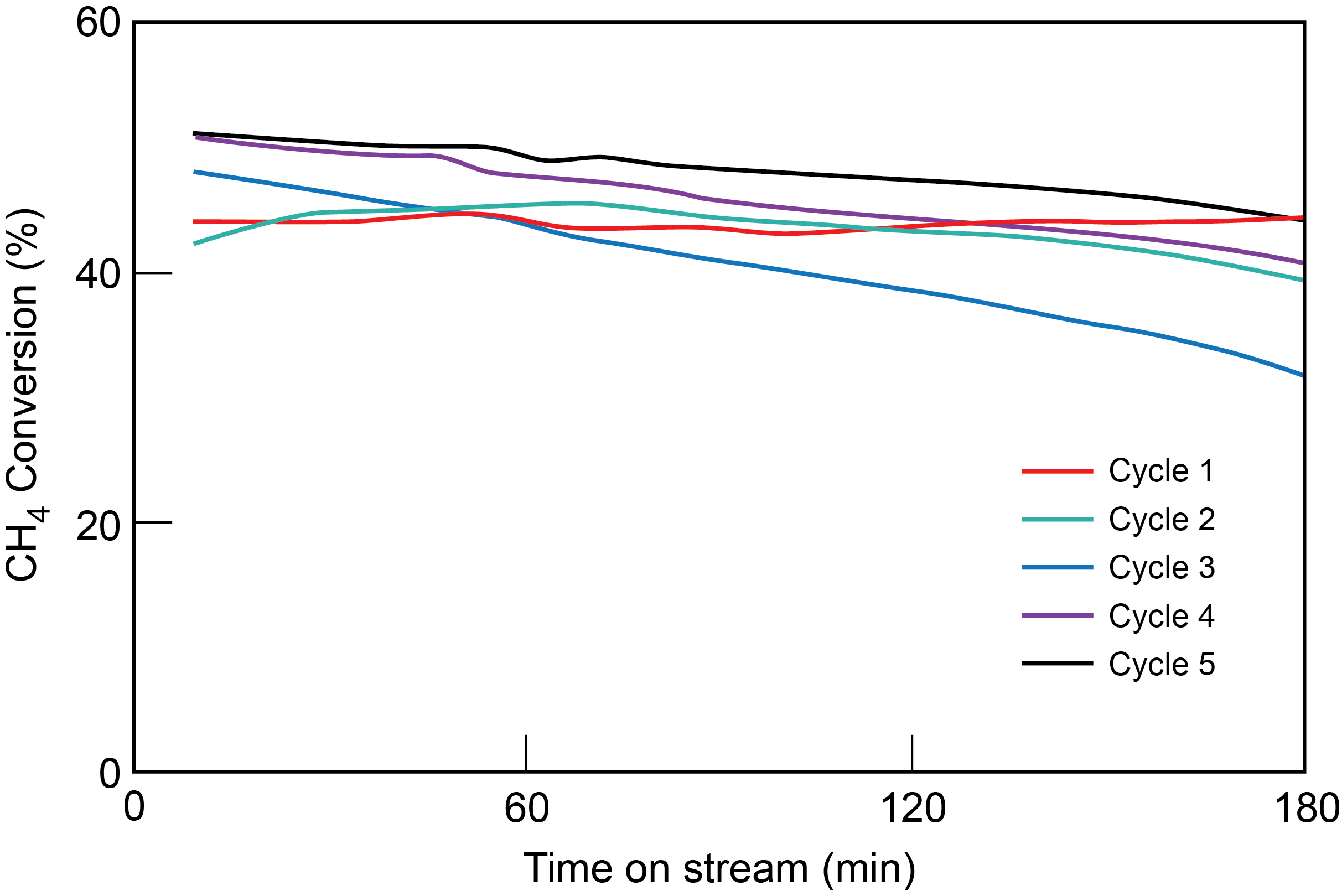
PNNL testing showed that through five TCD cycles, with each cycle lasting three hours, the bimetallic catalyst sustained reactivity and selectivity for producing both hydrogen and high-value solid carbon co-products.
Meanwhile, C4-MCP evaluated market opportunities for the crystalline carbon co-products, which have attractive properties, such as electrical conductivity, high tensile strength, and high thermal and chemical stability. The company projected the global market for the carbon nanotubes to increase from approximately $3.5 billion in 2016 to $8.7 billion by 2022, with robust growth rates of more than 17 percent annually.
"In the past, CNTs have faced headwinds in market adoption primarily due to price," said Will Kain, chief executive officer of C4-MPC. "CNTs produced through TCD, however, project to cut the prevailing market price by over 75 percent, which facilitates much more rapid prototyping and customer acceptance."
Based on process throughput and an estimated carbon price range between $0.7/kg - $1.2/kg, PNNL technoeconomic analysts project a net hydrogen production cost of $2.0/kg at 40,000 - 140,000 tons/year, assuming successful technology scale-up.
Also, conventional steam methane reforming typically emits around 11g of CO2 per g of hydrogen. For methane pyrolysis, PNNL projects a decrease in carbon dioxide emissions by 85 percent or more, depending on how the process is heated-for example, burning some of the produced hydrogen will result in zero emissions.
Phase two: scaling up to a fluidized bed reactor
Today, methane pyrolysis remains largely in the research phase because the gas isn't very reactive; this makes it hard to pyrolize-the hardest, in fact, according to Dagle. The conversion requires high operating temperatures and therefore a lot of energy.
Existing methane pyrolysis approaches are relatively inefficient at producing hydrogen and yield low-quality carbon products. The only known commercial methane pyrolysis technology in North America is a demonstration plant in Hallam, Nebraska.
The next phase of the TCD project would focus on scaling up the process for a pilot-scale demonstration in a fluidized bed reactor. Unlike a catalyst anchored to a fixed bed, the fluidized bed "bubbles," allowing the spent catalyst and carbon products to be continuously skimmed off. The spent catalyst can then be moved to another unit for resynthesis before restarting the cycle.
"It's kind of messy-there's a lot going on-but the fluidized bed enables us to have that separation and transport. It's a key step and that's what we have to scale up," said Dagle.
PNNL and WVU would also evaluate adjusting operating temperatures for higher yields per cycle, as well as ways to increase catalyst stability for more cycles before replacement. C4-MCP would look for new market opportunities made possible by the unique crystalline properties of the carbon co-products.
"It's kind of messy-there's a lot going on-but the fluidized bed enables us to have that separation and transport. It's a key step and that's what we have to scale up." - Rob Dagle, PNNL chemical engineer
Successful scale-up of the technology, combined with cost offsets from sales of carbon co-products, provides a pathway for fuel cell powered vehicles to close the cost gap with conventional gasoline and diesel fuels. The technology could help pave the way for one more right: carbon-free microgrid systems that move California closer to its ambitious emissions reduction goals.
Reference: Xu M., J.A. Lopez-Ruiz, L. Kovarik, M.E. Bowden, S.D. Davidson, R.S. Weber, and I. Wang, J. Hu, and R. Dagle. 2021. "Structure sensitivity and its effect on methane turnover and carbon co-product selectivity in thermocatalytic decomposition of methane over supported Ni catalysts." Applied Catalysis A: General 611. PNNL-SA-158733. doi:10.1016/j.apcata.2020.117967






