N. Hanacek/NIST
To effectively repel an enemy invasion, it helps to have accurate intelligence about that enemy's weaponry and attack plan. Medical scientists laboring to repel infectious viruses, such as those that cause COVID-19 and HIV, now have a better method for obtaining that intel because of research from the National Institute of Standards and Technology (NIST).
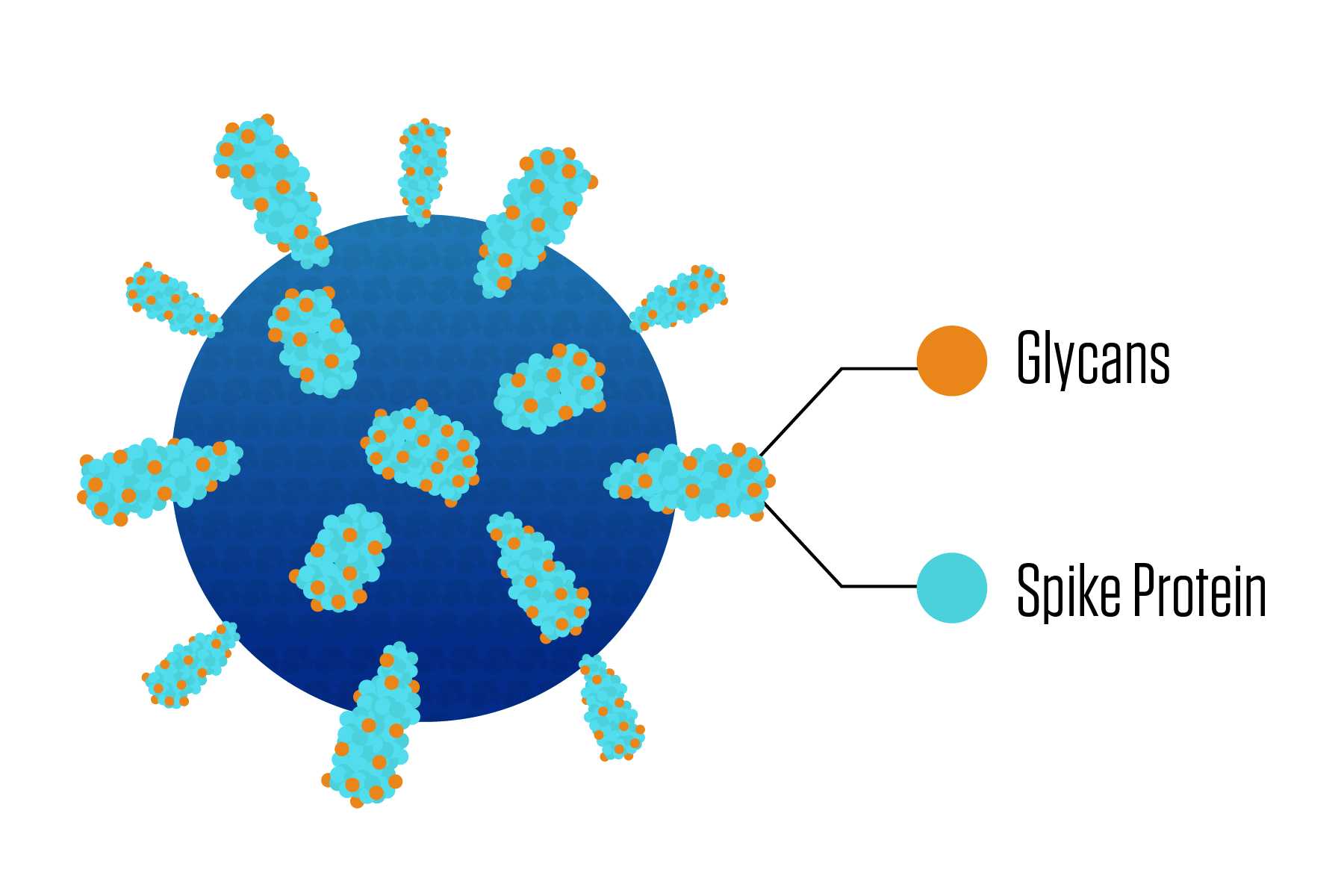
The chief weapon many viruses use to latch onto and invade a victim's cell is a spike-shaped protein jutting from the virus surface. Because the spike proteins play an essential role in the infection process, vaccines and treatment methods often target them, but these proteins are not easy marks.
One reason is that each spike protein is draped in a varied bunch of sugar molecules. These sugars help the virus particle both infect the cell and evade the immune system. Until now, our ability to determine the types of sugars at specific spots on the spike proteins has largely depended on educated guesswork.
The NIST team's new method dramatically improves the ability to identify these sugar molecules accurately. In a paper published in the Journal of Proteome Research, the team details the types, quantities and positions of the sugars branching from the spike proteins on the SARS-CoV-2 viral particle, which causes COVID-19. While the results will assist scientists in efforts to repel COVID-19 in particular, the findings' broader value lies in the method that revealed them, as applying it could improve the ability to repel a host of other viruses that plague humanity.
According to NIST's Stephen Stein, one of the paper's authors, characterizing the confusing array of sugars on the spike protein is crucial for giving medical science a clear picture of the enemy.
"If you're dealing with viral interactions at the molecular level, you must know everything about the molecule. The more you know, the more confident you'll be about how it works," said Stein, a chemist and NIST Fellow. "How does the particle evade the immune system's antibody attack? Answering that question requires details of the particle's molecular structure to be known, but until now this 'sugar coating' has not been known with precision. Any uncertainties in its structure will lead to uncertainties in unraveling its behavior."
Those limitations have made life difficult for other scientists who are trying to create ways to fight viral infection. Getting a detailed look at how the sugars behave could make a substantial difference to researchers like S. Saif Hasan, an assistant professor at the University of Maryland School of Medicine who studies how viruses invade our cells and transform them into virus-making factories.
"The NIST team's new method gives us unprecedented insight at the atomic level chemistry that underlies a successful infection," Hasan said. "It gives us new avenues to investigate the quality of vaccines against different viruses. This is something that has not been easily possible in the past."
Those different viruses are a rogue's gallery of human pathogens including COVID, HIV, influenza and Ebola. All of these particular viruses have spiky proteins that extrude outward from the surface.
Making the spike proteins all the more difficult to visualize are the different varieties of sugar molecules, known as glycans, that branch outward from the spikes. Scientists have lacked the ability to determine a spike's glycosylation profile - a map of which particular sugars sit where on a spike. A widely used device called a mass spectrometer can provide a good general rundown of all the glycans that appear in a sample, but until now, scientists have lacked a rigorous data analysis method that can turn this rundown into a map with trustworthy details.
The NIST team developed that analysis method over a painstaking stretch of months, using the mass spectrometer to look closely at samples of the SARS-CoV-2 spike protein obtained from 11 different vendors. The team members used their collective experience with determining and validating mass spectra to come up with entirely new sets of algorithms that can analyze a sample, and the multiple sample sources allowed them to make sure they were getting accurate results regardless of protein origin.
"It's no secret to the scientific community that the data analysis here is tough," Meghan Burke Harris, another author of this work, said. "Many glycans look similar, and they are often difficult to distinguish. But the extensive measurements and data analysis here provides a reliable method for this task - one that lays the groundwork for making accurate measurement."
In addition to the broader method, the team's paper offers a library of SARS-CoV-2 gycosylation profiles and includes visual representations of the results. Hasan said that their graphical form allows him to look at the data intuitively, potentially solving problems that would have been intractable.
"As a hypothetical example, let's say there are two batches of the same vaccine that were made separately. For some unknown reason, one provides better protection than the other," Hasan said. "With this technique, we could get more insight into why one batch is better than the other, and we could use that in the future to design better vaccines."
Paper: M.C. Burke, Y. Liu, C. Remoroza, Y.A. Mirokhin, S.L. Sheetlin, D.V. Tchekhovskoi, G. Wang, X. Yang and S.E. Stein. Determining Site-Specific Glycan Profiles of Recombinant SARS-CoV-2 Spike Proteins from Multiple Sources. Journal of Proteome Research. Aug. 30, 2023. DOI: 10.1021/acs.jproteome.3c00271






