Joint release by Hokkaido University and the University of Tokyo.
The discovery of a stable single-electron covalent bond between two carbon atoms validates a century-old theory.
Covalent bonds, in which two atoms are bound together by sharing a pair of electrons, form the scaffolding that underpins the majority of organic compounds. In 1931, the Nobel Laureate Linus Pauling suggested that covalent bonds made from just a single, unpaired electron could exist, but these single-electron bonds would likely be much weaker than a standard covalent bond involving a pair of electrons.
Since then, single-electron bonds have been observed, but never in carbon or hydrogen - the hunt for one-electron bonds shared between carbon atoms has stymied scientists.
Now, a team of researchers from Hokkaido University has isolated a compound in which a single electron is shared between two carbon atoms in a remarkably stable covalent bond, known as a sigma bond. Their findings are published in the journal Nature.
"Elucidating the nature of single-electron sigma-bonds between two carbon atoms is essential to gain a deeper understanding of chemical-bonding theories and would provide further insights into chemical reactions," explains Professor Yusuke Ishigaki, of the Department of Chemistry at Hokkaido University, who co-authored the study.
The single-electron bond was formed by subjecting a derivative of hexaphenylethane, which contains an extremely stretched out paired-electron covalent bond between two carbon atoms, to an oxidation reaction in the presence of iodine. The reaction produced dark violet-colored crystals of an iodine salt.
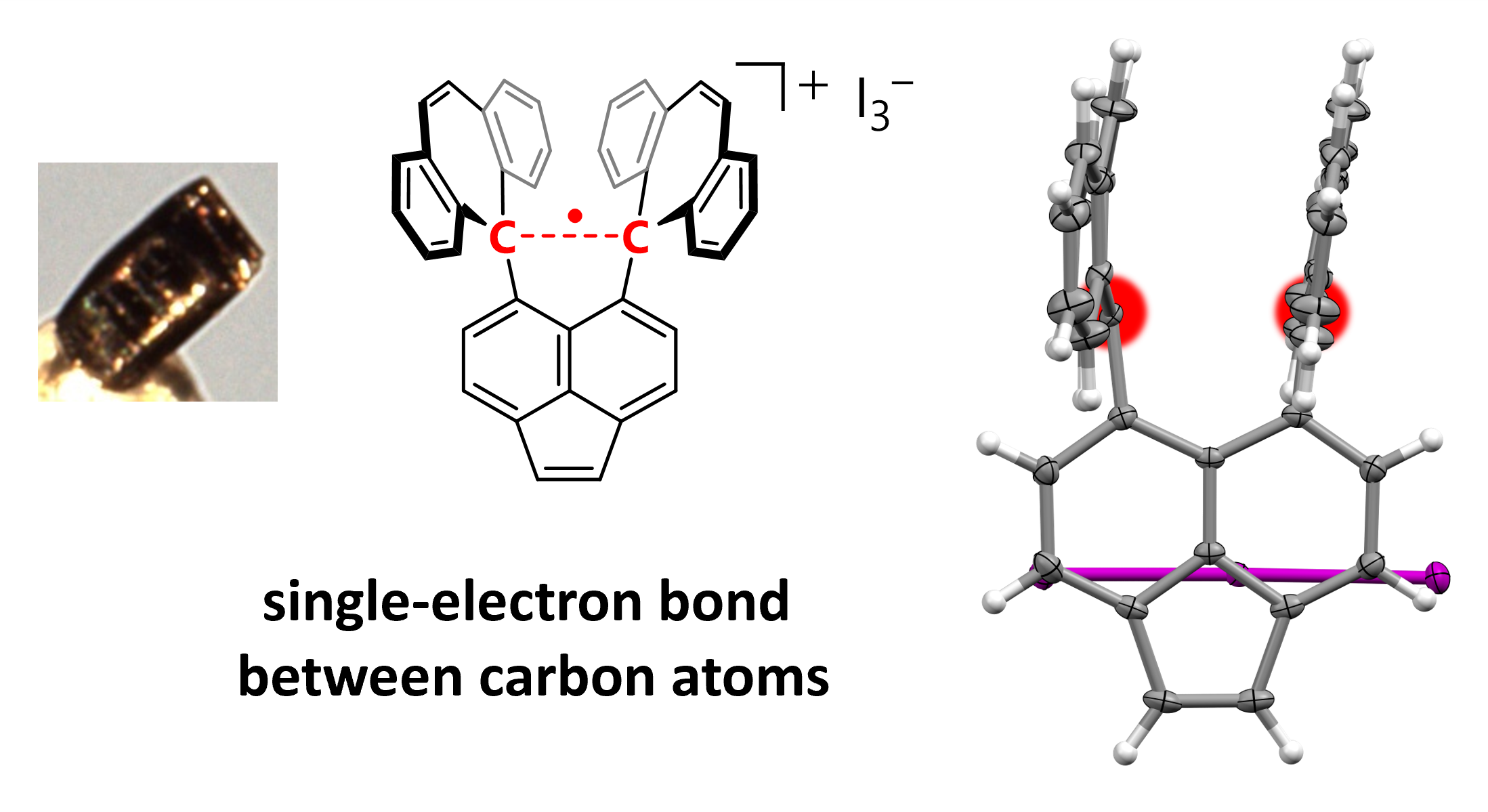
Structure of the compound highlighting the C-C sigma bond (red). (Takuya Shimajiri, et al. Nature. September 25, 2024)
The team used X-ray diffraction analysis to study the crystals and found that the carbon atoms in them were extremely close together, suggesting the presence of single-electron covalent bonds between carbon atoms. They were then able to confirm this using a form of chemical analysis called Raman spectroscopy.
"These results thus constitute the first piece of experimental evidence for a carbon-carbon single-electron covalent bond, which can be expected to pave the way for further developments of the chemistry of this scarcely-explored type of bonding," says Takuya Shimajiri, the lead author of the paper and now at the University of Tokyo.

(from left) Yusuke Ishigaki, Soki Kawaguchi, Takuya Shimajiri, and Takanori Suzuki of the research team. (Photo provided by Yusuke Ishigaki)
Original Article:
Takuya Shimajiri, et al. Direct evidence for a carbon-carbon one-electron σ-bond. Nature. September 25, 2024.
DOI: 10.1038/s41586-024-07965-1
Funding:
This work was supported by the Masason Foundation; the Research Program "Five-star Alliance" within "NJRC Mater. & Dev." of the Ministry of Education, Culture, Sports, Science and Technology (MEXT) of Japan; a Toyota Riken Scholarship; Grants-in-Aid from MEXT (Japan) and the Japan Society for the Promotion of Science (JSPS; JP23K13726, JP23K20275, JP23K21107, JP23H04011); and the Japan Science and Technology Agency (JST) PRESTO (No. JPMJPR23Q1).







