Dynamic structure of FLVCR proteins and their function in nutrient transport in our cells revealed
It is known that malfunctions of the proteins FLVCR1 and FLVCR2 lead to rare hereditary diseases in humans that cause motor, sensory and neurological disorders. However, the biochemical mechanisms behind this and the physiological functions of the FLVCR proteins have been unclear to date. An interdisciplinary team of researchers from Frankfurt am Main, Singapore and the USA has now deciphered the FLVCR proteins' 3D structures and their cellular functions. The researchers have shown that the proteins transport the cellular building blocks choline and ethanolamine. Their findings contribute significantly to understanding the pathogenesis of rare diseases and developing new therapies.
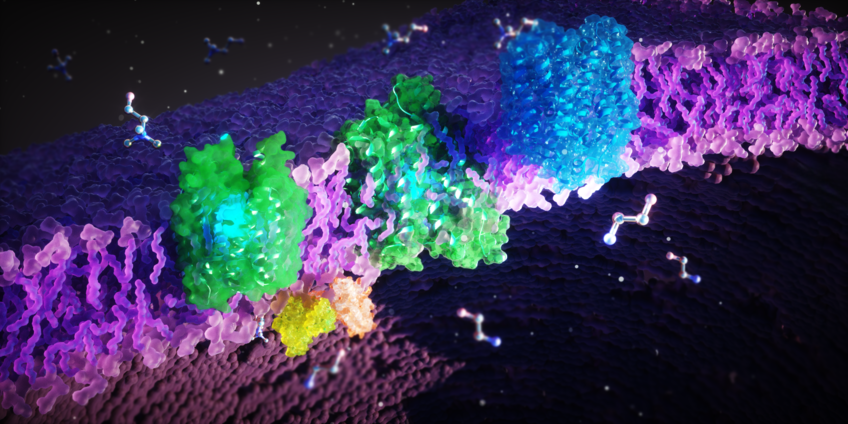
FLVCR proteins (green, blue) sitting in the cellular membrane (purple). These proteins transport the cellular building blocks ethanolamine and choline across the membrane.
© Ella Maru Studio
In hospital TV series such as Scrubs or Dr. House, medical doctors search for correct diagnoses and possible treatments for patients with sometimes puzzling or strange symptoms. In reality, this process often takes years for those affected by rare diseases. In many cases, there is no effective medication and therapeutic options are limited.
Approximately 6-8% of the world's population suffer from a rare disease. That's around 500 million people, even though each of the over 7000 different diseases only affects around one in 2000 people. Since these diseases are so rare, medical and scientific knowledge about them is limited. There are only a few experts worldwide and social awareness is lacking.
Unraveling the structure and function of proteins to understand diseases and develop therapies
An international team of researchers led by Schara Safarian, project group leader at the Max Planck Institute of Biophysics as well as independent group leader at the Fraunhofer Institute for Translational Medicine and Pharmacology ITMP, and the Institute of Clinical Pharmacology at Goethe University Frankfurt, has now investigated the structure and cellular function of two proteins, FLVCR1 and FLVCR2, which play a causal role in a number of rare hereditary diseases. The scientists have published their findings in the prestigious journal Nature.
Malfunctions of FLVCR1 and FLVCR2 due to gene mutations cause rare diseases, some of which result in severe visual, mobility and sensory disorders - such as posterior column ataxia with retinitis pigmentosa, Fowler's syndrome or sensory and autonomic neuropathies. The latter can, for example, lead to a complete loss of pain sensation. "In many diseases, including the rare ones, cellular structures in our body are altered and this leads to malfunctions in biochemical processes," says Schara Safarian. "In order to understand the development of such diseases and develop therapies, we need to know how these proteins are structured at the molecular level and what functions they perform in healthy cells."
FLVCR1 and FLVCR2 transport the cellular building blocks choline and ethanolamine
The scientists have discovered that FLVCR 1 and FLVCR2 transport the molecules choline and ethanolamine across the membranes of our cells. "Choline and ethanolamine are essential for important bodily functions. They support the growth, regeneration and stability of our cells, for example in muscles, internal organs and the brain," explains Safarian. "Furthermore, choline is involved in fat metabolism and detoxification by the liver. Our body also needs it to produce the neurotransmitter acetylcholine which is crucial for our nervous system and is needed by our brain to control the organs. So, you can imagine that malfunctions of the FLVCR proteins can cause severe neurological and muscular disorders."
The researchers used microscopic, biochemical and computer-assisted methods to investigate the FLVCR proteins. "We shock-froze the proteins and then observed them under an electron microscope," explains Di Wu, researcher at the Max Planck Institute of Biophysics and co-author of the study. "An electron beam penetrates the frozen sample and the interaction of the electrons with the material creates an image." The researchers take many individual images and process and combine them computationally to obtain high-resolution 3D structures of proteins. In this way, they were able to decipher the structures of FLVCR1 and FLVCR2 and see how they change in the presence of ethanolamine and choline. Computer simulations confirmed and visualized how the FLVCR proteins interact with ethanolamine and choline, and dynamically change their structure to enable nutrient transport.
Safarian summarizes: "Our findings pave the way for understanding the development and progression of rare diseases associated with the FLVCR proteins. In the future, patients may be able to benefit from new therapies that restore their life quality."






