A genomic study of hydrogen-producing bacteria has revealed entirely new gene clusters capable of producing large volumes of hydrogen.
A culture of Vibrio generating hydrogen by fermentation of formate. (Tomoo Sawabe)
Vibrionaceae are a family of marine bacteria famous for their bioluminescence, but also notorious for including the cholera pathogen among their number. Vibrionaceae have not been attractive subjects in biofuel production, but another unique feature of the family - their ability to generate large volumes of hydrogen gas by breaking down a substance called formate into carbon dioxide and hydrogen through fermentation - has now emerged as a potential solution for green energy.
Now, a team including Professor Tomoo Sawabe at the Faculty of Fisheries Sciences, Hokkaido University; Ramesh Kumar Natarajan at the National Institute for Interdisciplinary Science and Technology, India; and Fabiano Thompson at the Federal University of Rio de Janeiro, Brazil, have used genome sequencing to investigate the mechanism behind this biochemical feat. Their findings were published in the journal Current Microbiology.

Six species of Vibrio being tested for hydrogen production. The species belong to the Gazogenes (red) and Portersiae clades (yellow). (Photo: K. Sato)
The researchers examined all 16 known species of Vibrionaceae, which are often found in symbiotic relationships with deep-sea animals. They focused on the sequence and structure of the Hyf-type formate hydrogenlyase (FHL) gene cluster. The hydrogenase enzyme from this gene cluster catalyzes the breakdown of formate into hydrogen and carbon dioxide. This complex is also found in Escherichia coli, another bacteria which can generate hydrogen through fermentation, though in much smaller amounts than Vibrionaceae.
"These analyses reveal unexpected diversity of FHL gene clusters and relationships between gene clusters and function in hydrogen production ability," Sawabe explains.
The team discovered two new types of FHL gene clusters among Vibrionaceae, bringing the total number of FHL gene clusters in these bacteria to six. They suggest that this diversity in the structure of the cluster is the result of speciation among the Vibrionaceae as they adapted and evolved to occupy a range of ecological niches.
The study also found differences in hydrogen fermentation and production associated with the different FHL gene clusters. Vibrio tritonius, a marine species, and Vibrio porteresiae, found in mangrove-dwelling wild rice, showed the highest levels of hydrogen production, while Vibrio aerogenes and Vibrio mangrovi showed the lowest.
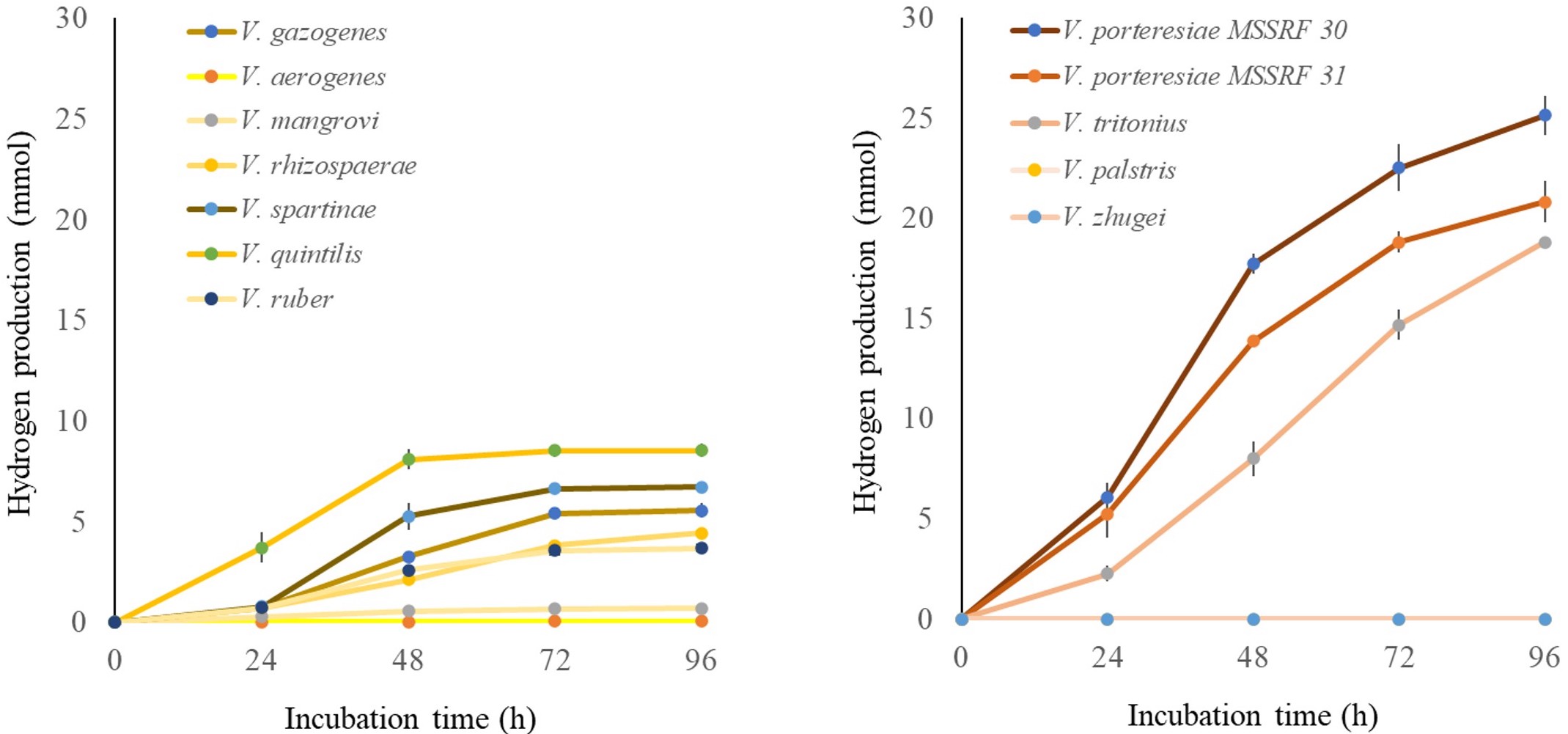
Hydrogen generation from the Gazogenes (left) and Portersiae (right) clades of Vibrio. Vibrio portersiae (dark red and dark orange lines, right graph) strains generate the largest amounts of hydrogen. (Tomoo Sawabe).
There was a correlation between levels of hydrogen production and how well the bacteria were able to take up formate back into their cells. "These genotypes strengthen formate metabolism as a possible key factor in maintaining fermentative hydrogen production in specific groups of vibrios," Sawabe concluded.
The findings support the researchers' proposal that some species were driven to evolve higher hydrogen production because of their need to detoxify formate from the environment - what they call the formate detoxification hypothesis. The findings could also shed light on the evolution of hydrogen fermentation in other bacterial species, such as E. coli.
Original Article:
Tomoo Sawabe, et al. Unexpected diversity in gene clusters encoding formate hydrogenlyase complex machinery in Vibrionaceae correlated to fermentative hydrogen production. Current Microbiology. March 25, 2025.
DOI: 10.1007/s00284-025-04176-3
Funding:
This research was supported by the Ministry of Education, Culture, Sports, Science and Technology of Japan (MEXT) KAKEN (JP16H04976, JP19H03041) and the Japan Society for the Promotion of Science (JSPS) Bilateral Research Project between Brazil and Japan (120200101).






